Sleep Monitoring System Overview
BM2000A Sleep Hypopnea Syndrome Monitoring
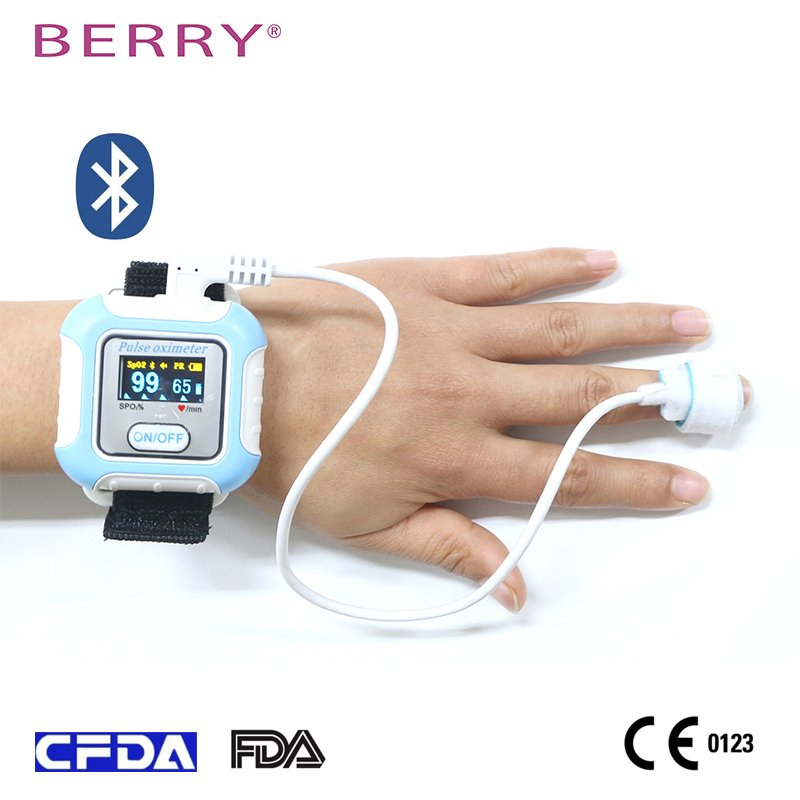
This Sleep Apnea Screening Monitor meets international standards including 510K, CE0123, ISO13485, FCC, and RoHS. Designed to accurately capture pulse signals and blood oxygen levels, it has been validated through extensive clinical trials to ensure medical-grade precision.
Key Features & Applications
Wide Application
Ideal for Aviation, Snoring monitoring, Pediatrics, Asthma, and COPD management.
- Specialized Monitoring: Optimized for sleep apnea and respiratory hypopnea monitoring.
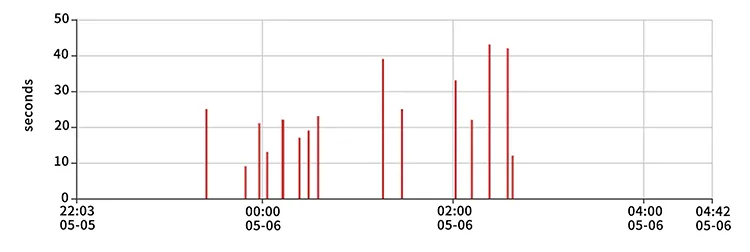
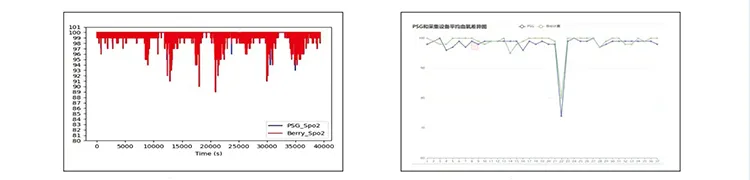
- Clinical Validation: Proven accuracy through comparative experiments with PSG (Polysomnography).
- Long Battery Life: 3.7V rechargeable lithium battery lasts for over 20 hours of continuous use.
- Universal Compatibility: Includes various sensors suitable for infants, children, and adults of all skin tones.
- Smart Connectivity: Connects via Bluetooth to generate comprehensive overnight sleep reports.
Operation Guide
01Download the Smart Health APP from Google Play or Apple Store.
02Wear the equipment for at least 6 hours during sleep.
03Wrap the foam SpO2 sensor securely around the fingertip.
04Data is transferred automatically via Bluetooth to your phone.
05Review and share your generated health reports.
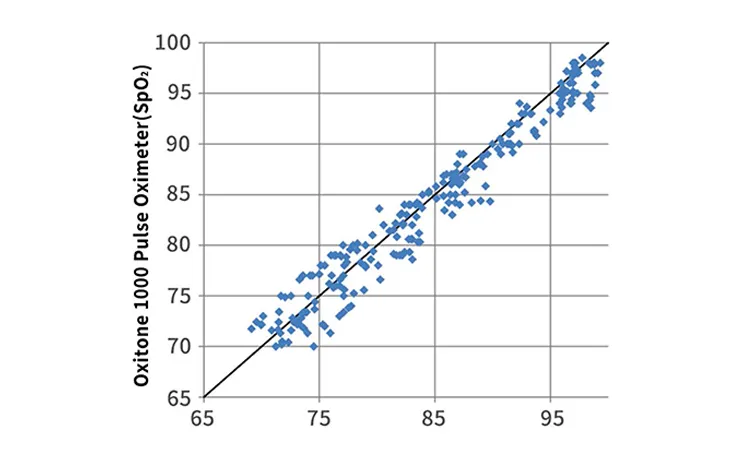
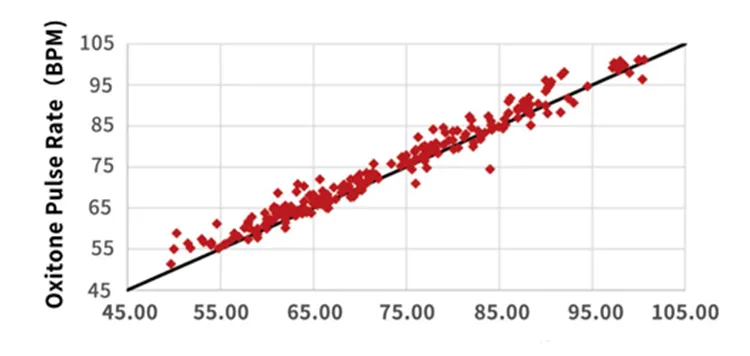
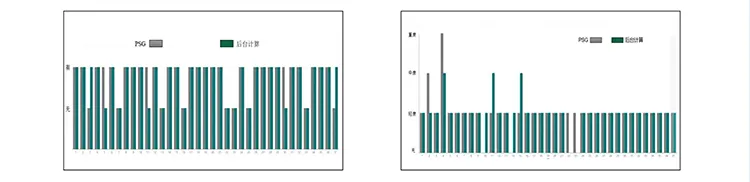
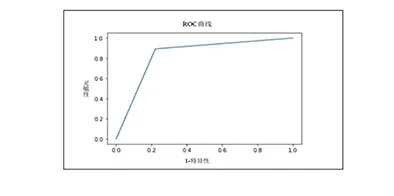
Accuracy & Clinical Performance
Correlation=0.98, P-value < 0.0001 compared to ECG Heart Rate.
| Clinical Trial Results Analysis |
| Category |
Sensitivity |
Specificity |
Accuracy |
| Hypoxemia Diagnosis |
89.29% |
77.78% |
92.59% |
| OSAHS Judgment |
94.17% |
61.21% |
89.62% |
| Mean SpO2 |
Average Calculation Accuracy: 98.51% |
Frequently Asked Questions
Who is the BM2000A sleep monitor suitable for?
The device is designed for monitoring sleep apnea and hypopnea syndrome. It is suitable for infants, children, and adults, and is widely used in aviation, asthma management, and COPD care.
How long can I use the device on a single charge?
The 3.7V rechargeable lithium battery is designed for low power consumption and can last for more than 20 hours of continuous monitoring.
Is the device medically certified?
Yes, the BM2000A has obtained international certifications including CE0123, 510K (FDA), and ISO13485, meeting strict medical quality standards.
How do I access my sleep reports?
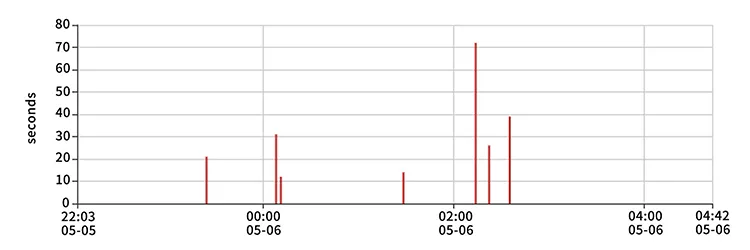
The device connects to the Smart Health App via Bluetooth. It records data every second and generates a detailed overnight report that can be viewed and shared directly from your smartphone.
What sensors are included in the package?
The standard configuration includes three types of probes: adult, child, and neonate, ensuring it can be used for patients of all ages.
What is the accuracy level of the blood oxygen measurement?
Clinical trials show a blood oxygen concentration accuracy of <2%, which exceeds the 510k requirement of <3%. The correlation with ECG heart rate is 0.98.