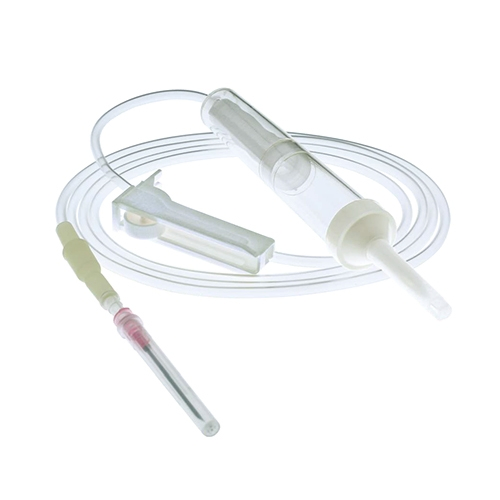










| MATERIAL | ABS, PVC, PP, RUBBER, STAINLESS STEEL |
| PACKING | INNER PE BAG, OUTER CARTON |
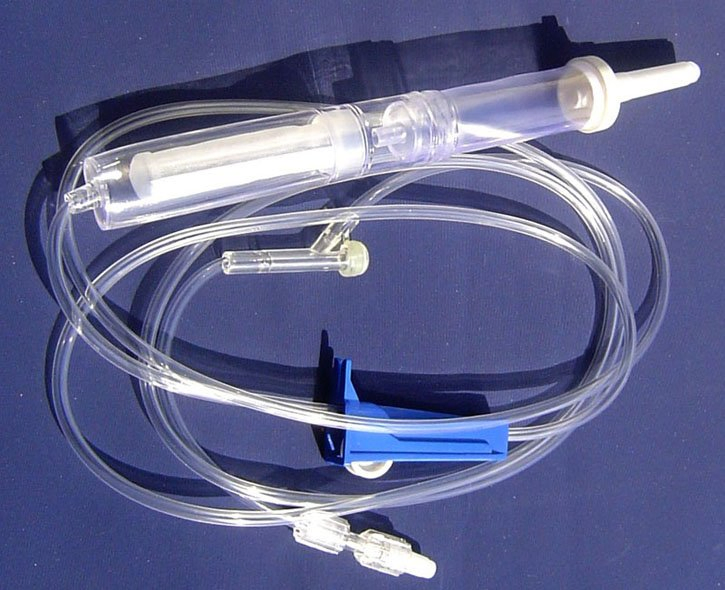
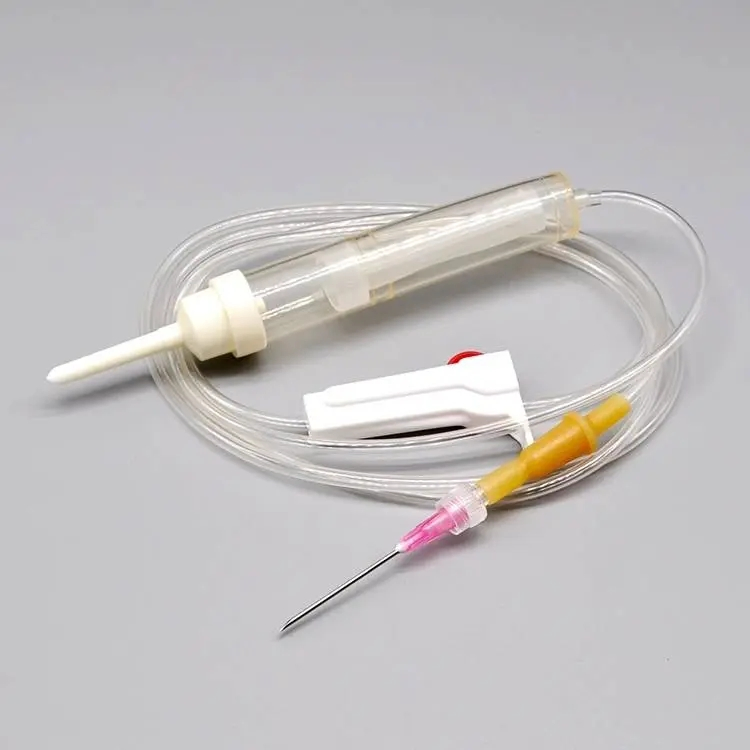
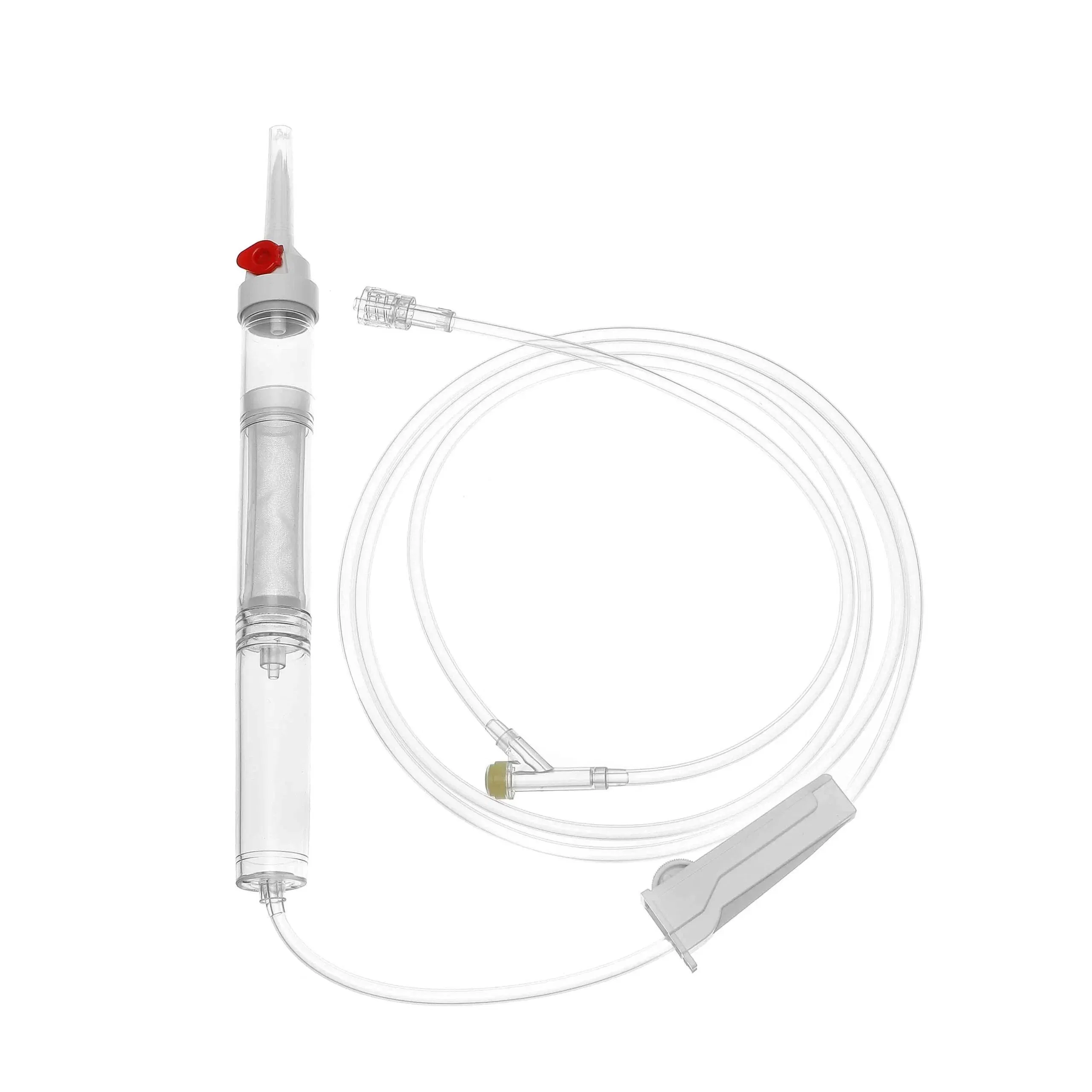
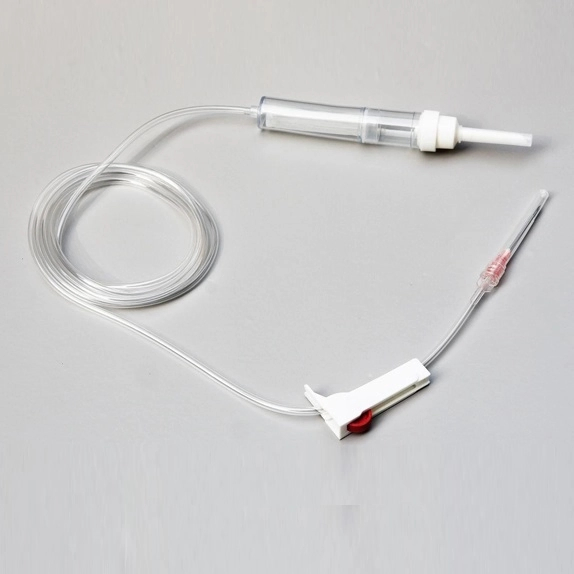
| COMPONENTS | SEE BELOW COMPONENT DETAILS |
| DIMENSION | 150CM-200CM |
| LOADING PORT | SHANGHAI PORT, NINGBO PORT, QINGDAO PORT |


USE Primary purpose is to keep the spikes clean and safeguard them from contaminants.
MATERIAL Made of medical grade PP or PE.
USE Designed to effortlessly pierce the bottle stopper of the infusion bag or bottle.
MATERIAL Made of ABS, which is rigid, durable, and resists chemicals.
USE Allows for flexibility in usage and prevents microorganisms from entering the infusion line.
MATERIAL PVC Cap (Blue) and PP Filter (White).
USE Soft, transparent chamber to collect fluid and act as a vital indicator of flow rate.
MATERIAL Normal PVC or DEHP-Free PVC.
USE Removes unnecessary particles and debris before entering the patient's bloodstream.
MATERIAL ABS shell and PP filter.
USE Includes roller clamp or microflow regulator to manage infusion speed.
MATERIAL ABS, PE, or POM.
USE Connects needle to tubing (Luer lock/slip). Hypodermic needle for skin penetration.
MATERIAL ABS connector; PP hub; Stainless steel cannula.