

1 / 5










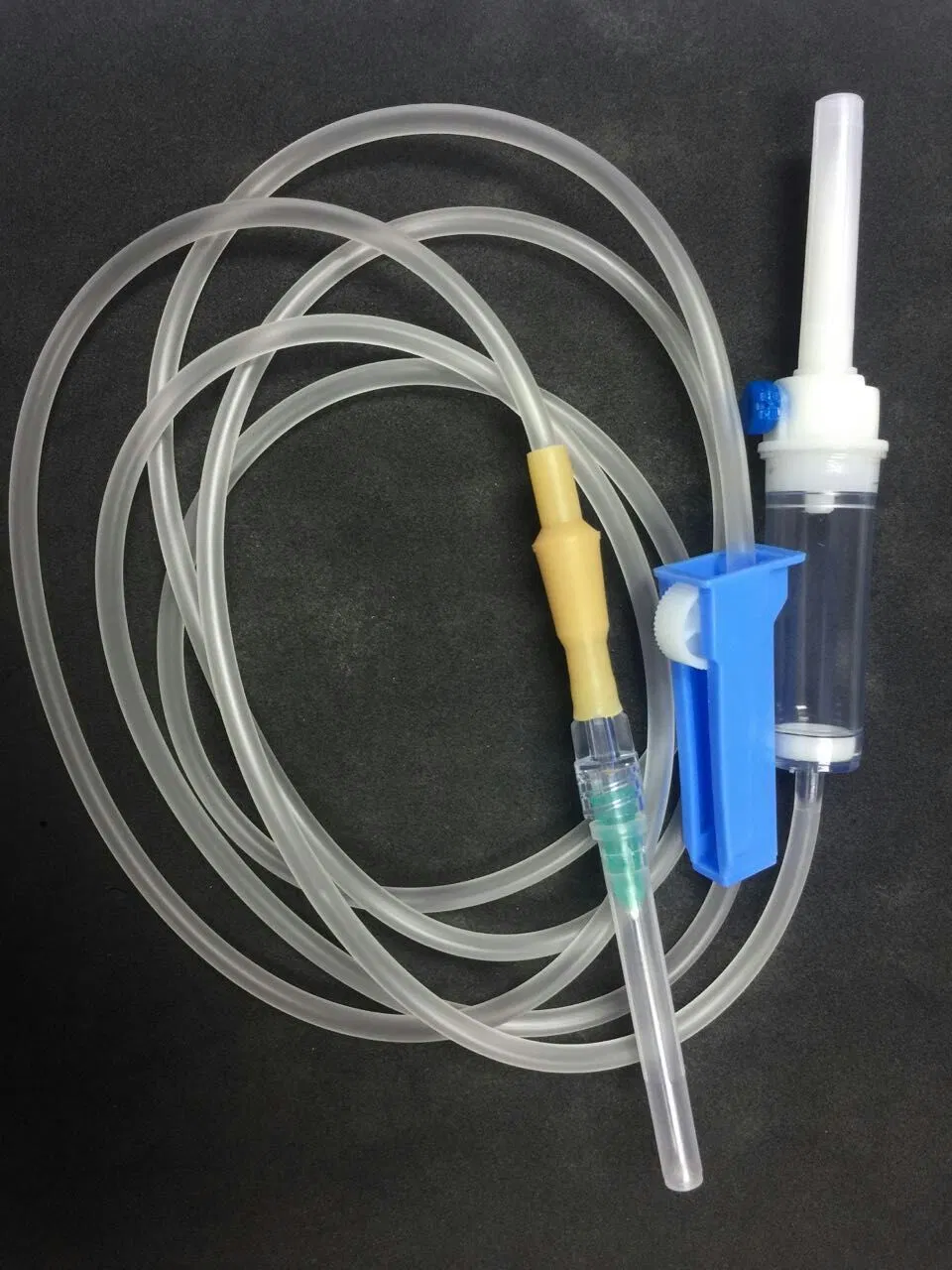
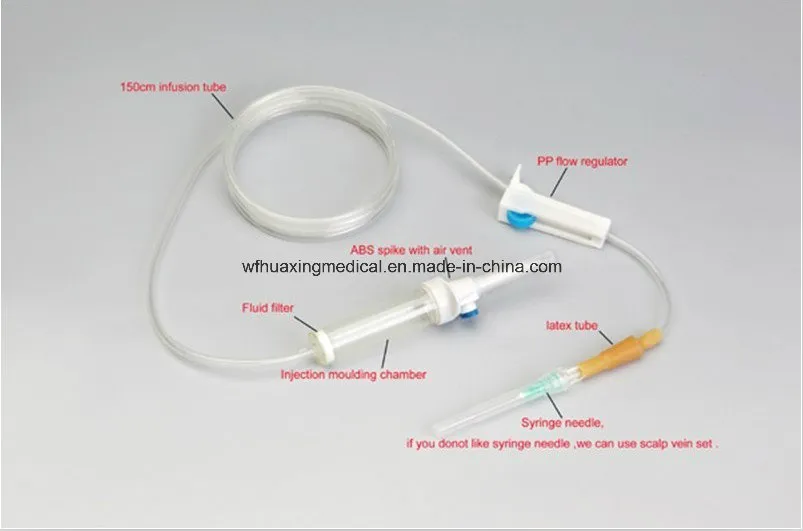
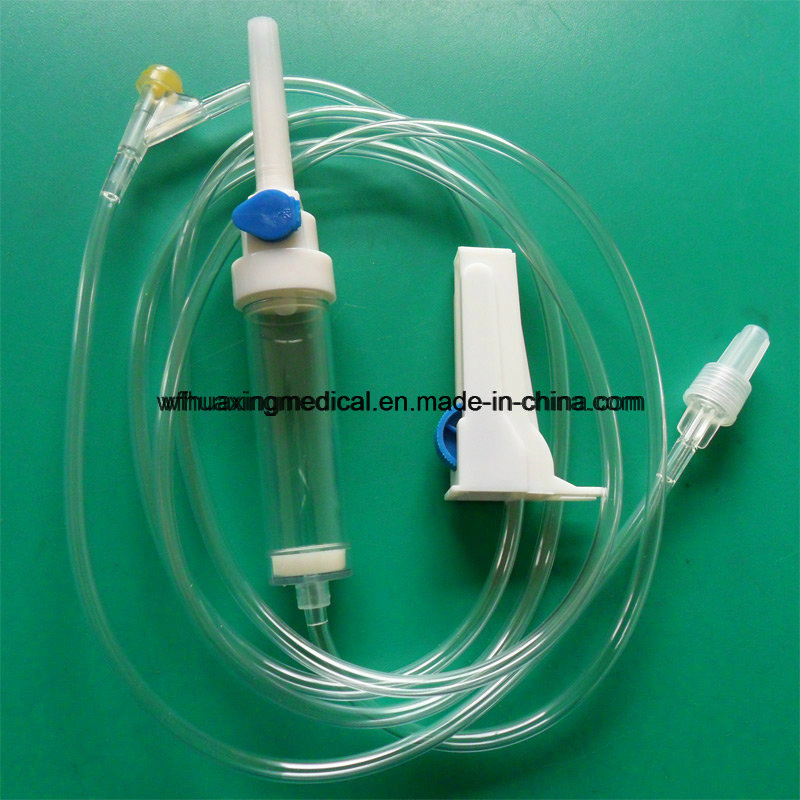
Vented spike, Dripping Chamber, Medicine Filter, Flow Regulator, Latex Tube, Luer Lock/Slip Connector, Infusion Tube, Y-injection port, Mould Chamber, etc.
| Commodity | Sterile infusion set with needle |
| Material | PVC tube, PE chamber |
| Sterile | Sterile by EO gas, non-toxic, non-pyrogenic |
| Type | 20 drops or 60 drops/ml |
| Tube Length | 150cm, 180cm, 200cm |
| Certificate | CE & ISO 13485 |
| Application | Intravenous infusion for human body |
| Shelf Life | Disposable, Valid for five years |