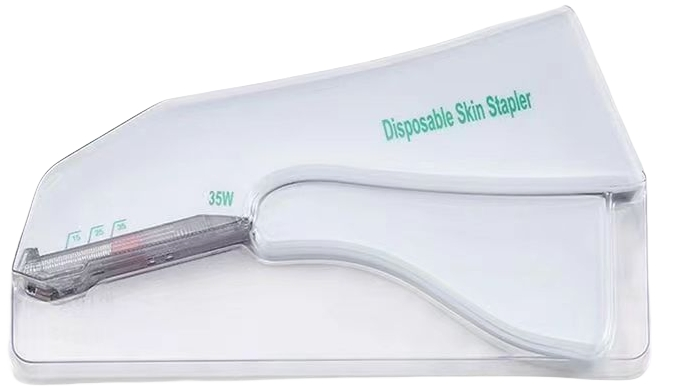














| Type | Surgical Skin Stapler |
| Colour | White |
| Model | 15W, 25W, 35W, 15R, 25R, 35R, 15H, 25H, 35H |
| Sterilization | Ethylene Oxide |
| Certificates | FDA, FSC |




Our company specializes in the production of medical consumables, established with a registered capital of 30 million. The main products are absorbable surgical suture with needle (Plain / Chromic Catgut, PGA, PDO, PGLA, PGCL, etc.), non-absorbable surgical suture with needle (Silk, Nylon, Polyester, Polypropylene etc.), suture materials, cassette sutures and surgical needle, lifting threads for beauty, medical facial mask, orthopedic casting tapes & splints, plaster, IV cannula, polypropylene meshes and absorbable bone nails and so on.
We have approved ISO13485 international quality system certificate, CE and FDA certificates. We have a large catgut plant and serve as a core enterprise of surgical absorbable sutures characteristic industrial base. The products are exported to Asia, Europe, Latin America, Africa, Middle East and other more than 70 countries and regions all over the world.


